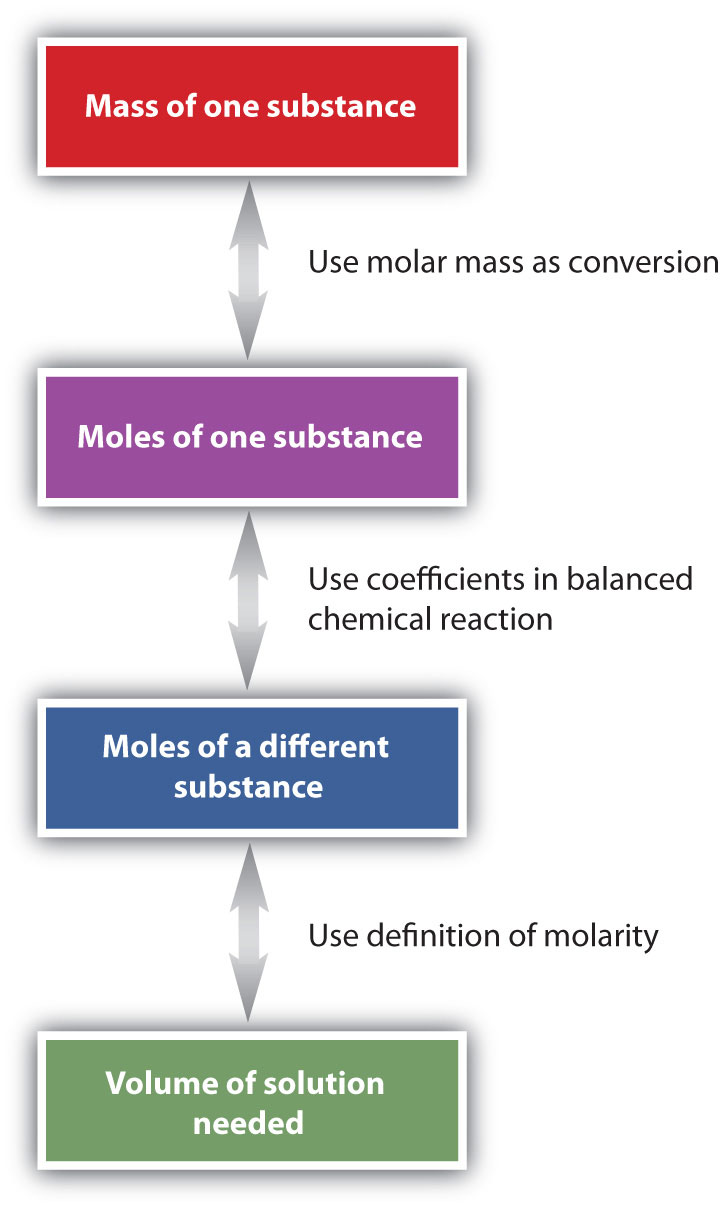
Since from the question you know the concentration of the sulfuric acid you need, then it follows that you must decide on what volume you need. Suppose that in one of your general chemistry experiments, you are required to use 2 M sulfuric acid (H 2 SO 4 ) for a certain reaction, but your lab instructor gave you 5 M H 2 SO 4. Now, let’s used it to answer the following question: To use the dilution formula, you must know at least three of the four variables in it. Note that if you report the concentration of solution as 2 M, the uppercase M is often used to represent the unit (mol/L), which is the unit for concentration reported in Molarity. If we translate this into a mathematical expression, we will get:Ĭalculating the concentration of dilute solutionĪs you may have noticed, the formula we just derived is the general formula for diluting a concentrated solution to a solution of lower concentration.

Volume (V) of concentrated solution times Molarity (M) of concentrated solution is equal to the volume (V) of dilute solution times the Molarity (M) of dilute solution. Moles of concentrated solution = moles of dilute solutionīut since we know that the amount of the coffee solution in moles is equal to the volume of the solution multiplied by its concentration in Molarity, then we can write that: If we translate the previous statement into a mathematical expression, we will get:
HOW TO CALCULATE PPM WITH MOLARITY FREE
This free PPM to Molarity calculator used for measurements of drinking water quality and producing chemical solutions and also use the above free formulas and examples to make your problems easy.A stock or standard solution is a solution in which you accurately know its concentration. Step 3: The calculator will display an answer on the screen. Step 2: Enter the value of molar mass in the second required input. Step 1: Enter the value of ppm in the first required input. Solution molarity = (70 * 0.001) / 33 molarity = 0.0021 mol/l Steps to use the PPM to Molarity calculator To find molarity = ? Molarity = ((ppm * 0.001) / molar mass) Given data ppm value = 70 ppm molar mass = 33 g/mol And, 33 g/mol is the atomic weight for calcium ion. Molarity = ((ppm * 0.001) / molar mass) ExampleĬalculate the molarity (mol/L) if there is 70 ppm of Ca2+ ions.

To convert ppm to molarity we can divide the ppm to 1000. On the other hand, molarity tells us that in one liter of water how many moles of molecules present. PPM stands for parts per million is a unit that is used in several branches in different ways.
